
IAEA-TECDOC-1430
Radioisotope handling
facilities and automation of
radioisotope production
December 2004

IAEA-TECDOC-1430
Radioisotope handling
facilities and automation of
radioisotope production
December 2004
The originating Section of this publication in the IAEA was:
Industrial Applications and Chemistry Section
International Atomic Energy Agency
Wagramer Strasse 5
P.O. Box 100
A-1400 Vienna, Austria
RADIOISOTOPE HANDLING
FACILITIES AND AUTOMATION OF RADIOISOTOPE PRODUCTION
IAEA, VIENNA, 2004
IAEA-TECDOC-1430
ISBN 92–0–116104–2
ISSN 1011–4289
© IAEA, 2004
Printed by the IAEA in Austria
December 2004
FOREWORD
If a survey is made of the advances in radioisotope handling facilities, as well as the technical
conditions and equipment used for radioisotope production, it can be observed that no fundamental
changes in the design principles and technical conditions of conventional manufacture have happened
over the last several years. Recent developments are mainly based on previous experience aimed at
providing safer and more reliable operations, more sophisticated maintenance technology and
radioactive waste disposal.
In addition to the above observation, significant improvements have been made in the
production conditions of radioisotopes intended for medical use, by establishing aseptic conditions
with clean areas and isolators, as well as by introducing quality assurance as governing principle in the
production of pharmaceutical grade radioactive products. Requirements of the good manufacturing
practice (GMP) are increasingly complied with by improving the technical and organizational
conditions, as well as data registration and documentation.
Technical conditions required for the aseptic production of pharmaceuticals and those required
for radioactive materials conflicting in some aspects are because of the contrasting contamination
mechanisms and due consideration of the radiation safety. These can be resolved by combining
protection methods developed for pharmaceuticals and radioactive materials, with the necessary
compromise in some cases.
Automation serves to decrease the radiation dose to the operator and environment as well as to
ensure more reliable and precise radiochemical processing. Automation has mainly been introduced in
the production of sealed sources and PET radiopharmaceuticals. PC controlled technologies ensure
high reliability for the production and product quality, whilst providing automatic data acquisition and
registration required by quality assurance. PC control is also useful in the operation of measuring
instruments and in devices used for packaging, identification and labeling.
This TECDOC summarizes major advances in the conditions of radioisotope handling facilities
ensued with relation to the reliability, quality assurance, aseptic processing and automation. Examples
of the major radioisotope production technologies are given together with several pictures of the
devices discussed. Also included are a few papers submitted by the experts who attended the
consultants meeting.
The IAEA wishes to thank the experts for valuable work and scientific contribution. Special
thanks are due to L. Baranyai from the Institute of Isotopes, Hungary for the final compilation of the
TECDOC. The IAEA officers responsible for this publication were D.V.S. Narasimhan and
M.R.A. Pillai of the Division of Physical and Chemical Sciences.
EDITORIAL NOTE
This publication has been prepared from the original material as submitted by the authors. The views
expressed do not necessarily reflect those of the IAEA, the governments of the nominating Member
States or the nominating organizations.
The use of particular designations of countries or territories does not imply any judgement by the
publisher, the IAEA, as to the legal status of such countries or territories, of their authorities and
institutions or of the delimitation of their boundaries.
The mention of names of specific companies or products (whether or not indicated as registered) does
not imply any intention to infringe proprietary rights, nor should it be construed as an endorsement
or recommendation on the part of the IAEA.
The authors are responsible for having obtained the necessary permission for the IAEA to reproduce,
translate or use material from sources already protected by copyrights.
CONTENTS
1. INTRODUCTION............................................................................................................................... 1
2. FACILITIES FOR HANDLING RADIOISOTOPES......................................................................... 2
3. ASEPTIC CONDITIONS IN THE PRODUCTION OF RADIOPHARMACEUTICALS .............. 15
4. EQUIPMENT AND DEVICES FOR USE IN HOT CELLS............................................................ 20
5. AUTOMATION, MECHANIZATION AND PC CONTROLLED PROCESSING ........................ 28
6. EXAMPLES OF MAJOR RADIOISOTOPE PRODUCTION SYSTEMS...................................... 30
BIBLIOGRAPHY ..................................................................................................................................41
PRESENTATIONS
Automation systems for radioisotope laboratories............................................................................ 45
D. Novotny
Advances in radioisotope handling facilities and automation of radioisotope production ............... 51
L. Baranyai
The present status of hot cell designs at NECSA.............................................................................. 59
M. Smith
LIST OF PARTICIPANTS ................................................................................................................... 65
1. INTRODUCTION
In order to handle radioactivity, special facilities are required to shield the radiation emitted and
to prevent contamination of the environment by the radioactive materials released during handling and
processing. In production laboratories the processed activities are high and therefore the requirements
for shielded facilities with well controlled ventilation and remote handling devices are greater. Several
versions of shielded hot cells and remote handling devices have been developed and used over the
years in major radioisotope production centres. The evolution of different designs has reflected the
growing emphasis on optimization of protection and safety. As radioisotopes are increasingly being
used in various fields, the demand for larger quantities has risen and more complex handling facilities
are in need in the isotope production laboratories.
Some of the recent developments in the use of radioisotopes in medical field have also
significantly impacted on the evolution of handling facilities. Application of pharmaceutical good
manufacturing practice (GMP) requirements for air quality and processing conditions in the handling
facilities of radioactive pharmaceuticals has led to significant improvements in the construction of
isolator-like hot cells and clean rooms with HEPA filtered ventilation and air conditioning (HVAC)
systems. Clean grade A (Class 100) air quality isolator-like hot cells compliant with GMP
requirements for handling radiopharmaceuticals are now available commercially. Nevertheless, the
application of clean room requirements in radioisotope laboratories in general and hot cells in
particular is technically not an easy task. The technical problems have not been completely overcome
and need on-going efforts.
Furthermore, GMP requirements exclusively designed to conditions for producing radioactive
pharmaceuticals should be elaborated and distributed to facilitate the design, construction and
operation of such facilities. GMP guidelines are guidelines for radioactive pharmaceuticals, by
describing their specific character and refer to the production of sterile pharmaceutical products.
Due to the beneficial nuclear and chemical character of
99m
Tc radionuclide as well as its easy
availability from the
99
Mo-
99m
Tc generator, it is the ‘workhorse’ of the nuclear medicine and is widely
used in nuclear medicine centres for diagnostic purposes. The manufacture of
99
Mo-
99m
Tc generators
ranges from small scale batch techniques through semi-automatic dispensing systems to fully
automatic production lines. Their manufacture and quality control are also made under aseptic
conditions to comply with GMP requirements.
The last decade has seen an increased use of positron emission tomography (PET) in regular
diagnostic imaging and PET radiopharmaceuticals, (particularly
18
F-FDG in the hospitals). The 511
KeV, high-energy radiation needs thicker shielding and more sophisticated handling devices. In view
of the short half-lives, the emphasis is also increasingly on the process and handling as per GMP than
final QC. The clean area concept is more often applied to isolator-like hot cells for PET
radiopharmaceuticals. The need for rapid, remote and reliable synthesis of PET radiopharmaceuticals
has also been responsible for the introduction of microprocessor controlled synthesis modules. This
experience has also led to the development of similar microprocessor controlled synthesis systems for
other radioactive pharmaceuticals.
Miniature size radioactive sealed sources of
192
Ir,
125
I and
103
Pd have found widespread
applications in brachytherapy of cancer. The production of such sealed sources together with that of
the conventional sealed sources (
192
Ir and
60
Co) also require remote precision welding using laser or
arc and microprocessor controlled positioning devices.
Several radioisotope production centres have developed innovative devices and systems for
remote handling of various important operations in hot cells such as target handling, capping and
decapping, dispensing and autoclaving. Often these developments have resulted in significant
reduction of radiation dose and increased the GMP compliance.
1
These developments in the radioisotope handling technology are of considerable practical value
to radioisotope production laboratories in several Member States. Compiling the salient points of these
developments in a technical report is expected to make the information readily available to them for
reference. With this objective, a consultants meeting was held in Pretoria, South Africa from 17 to 20
February 2003 to review the recent developments in the automation and remote handling technologies
as well in the aseptic production conditions of radioactive pharmaceuticals.
The topics for discussion in the above meeting included the description of radioisotope handling
facilities, hot cell designs, devices specifically designed for hot cell operations, aseptic production
conditions (clean rooms and isolators), PET synthesis modules,
99
Mo-
99
Tc
m
generator production lines,
technological equipment, microprocessor controlled technologies, automation in sealed source
production, and batch processing in miniature sources.
This TECDOC gives the most recent information collected on the development of radioisotope
handling facilities, process equipment and devices as well as automation of the production. The
dissemination of these information may assist pharmaceutical grade radioisotope producers to design
modern facilities complying not only with requirements relating to radioactive materials but also
providing aseptic conditions for the production of radioactive pharmaceuticals. Automation in the
radioisotope production enables producers to reduce radiation dose, to increase reliability of the
systems and to improve quality of products.
This technical report consists of two sections. The first part summarizes the discussions of the
above topics, ending with conclusions/recommendations by the participants. The second is a
compilation of a few papers covering the experiences in the consultants’ institutions.
This report is illustrated with several photos of the facilities and devices provided by the
consultants.
2.
FACILITIES FOR HANDLING RADIOISOTOPES
Due to the radiation emitted from radionuclides and the risk of radioactive contamination, the
radioactive materials are potentially hazardous to their environment. Handling and processing facilities
are therefore specially constructed to minimize radiation exposure.
Based on the risk of incorporation, radioisotope laboratories have been classified in some
countries into C, B and A categories. As large quantity of activity is handled, radioisotope production
facilities belong to the ‘A’ category. Such facilities must be designed for the safe handling of
radioactivity with respect to personal safety and safety of the surroundings. This safety consideration
should include the building’s safety interlock system, surveillance equipment and radiation
monitoring.
A radioisotope production facility is preferably constructed as a single storey building to
remove any doubt whether the top floor can carry the weight of shielding. The general finish of the
production laboratories need to be smooth; and corners between walls and floors are preferably
rounded to ease decontamination. Doors and windows are designed to provide increased sealing.
A well planned ventilation system forms the basis of contamination control in a radioisotope
laboratory. For this purpose filtered air has to be supplied to the laboratory and exhausted. Hot cells
need to be provided with a separate exhaust system. Direction of flow of air within the laboratory
needs to be from the zones with the lowest levels of radioactivity towards the zones with the highest
potential levels of radioactivity. In practice, this is arranged by supplying fresh air to the corridors.
This air is drawn through the production laboratories into the boxes and finally filtered before being
exhausted to the atmosphere.
2

As radioisotope handling and processing centres are generally located not within or close to big
cities or industrial areas, the air is usually clean enough to be fed into the laboratory through
inexpensive, low grade filters. To cover the possibility of failure in the ventilation system, a warning
system need to be fitted.
Change rooms are incorporated to divide the radioactive zone from the inactive area. A full-
body radiation detector gate is placed between the two zones to check potential personal
contamination before leaving the radioactive zone (Fig. 1). Consideration needs to be given to the
provision of an emergency electric generator to be used, in the event of a main power failure.
FIG. 1. Radiation control gate for checking personal contamination
(Source: IZOTOP, Hungary).
In order to prevent the uncontrolled spread of radioactive contamination, the processing of
radioactive materials requires an exhausted and shielded special enclosure called hot cell. Hot cells are
arranged in the production laboratory in series, in blocks or individually with provision accessibility to
maintenance (Fig. 2).
3

FIG. 2. Highly shielded hot cells for extremely high activities
(Source: NTP, South Africa).
Radioactive materials are produced typically in small batch sizes using materials in small
quantities. Heavy lead containers are used for protection from radiation and hence there is a need for a
crane to move these containers within the area. Attention must be paid to radioactive waste disposal.
Discharged liquid wastes are to be monitored and treated while solid wastes are disposed at authorized
radioactive waste repositories.
Control devices placed in the front of hot cells serve for operating the technological equipment
and measuring instruments (Figs 3–4).
4

FIG. 3. Hot cell series with remote handling tongs
(Source: IZOTOP, Hungary).
FIG. 4. Front of a hot cell with remote handling tongs
(Source: IPEN, Brazil).
The floor of the containment box is where most operations are performed and its use need be as
effective as possible. Large items, such as autoclaves, ion-chambers storage containers and reaction
vessels are sometimes better placed under the floor. Other than space saving, a further benefit could be
the removal of services and their feed-through connectors from the box. Additionally these items can
be shielded from the box or alternatively the box could be shielded from them. This may be done
singularly, as in the case of the ion-chamber, or collectively where the whole floor of the box (and
below box walls) is shielded. Storage of waste under a lead slab reduces the radiation damage to in-
cell equipment and can reduce radiation to the operators (Fig. 5).
5

FIG. 5. Solid and liquid radioactive waste disposal equipment
(Source: IPEN, Brazil).
In order to avoid cross-contamination individual hot cells or blocks of hot cells are preferred to
whole series of hot cells used for different technologies. Separation of the front and rear of the hot
cells from access and air supply point of view is is one of the options to provide better radiological
protection.
The general facilities for radioactivity handling in conventional radiochemical laboratories
including hot cells have not undergone dramatic changes. Major new developments are installation of
aseptic production areas (clean rooms) within radioactive laboratory for the production and testing of
pharmaceutical grade radioactive products.
HOT CELL DESIGNS
Hot cells are exhausted and shielded enclosures equipped with remote handling tongs or master-
slave manipulators for processing radioactive materials. Application of hot cells is based on the
principle that protection is the cheapest if containment is closest to the radiation source.
Conventional hot cells
Hot cells are generally made of ordinary mild steel covered by a good quality epoxy resin paint
or made of stainless steel. Operations are carried out through one of the larger faces by using
manipulators. Connections for services are welded into the bottom of the box or service lines may be
brought in through a panel in the top face. An accessible door at the back provides access for
maintenance and cleaning (Fig. 6).
Hot cells have to be shielded against radiation from gamma emitters for which a rigid support
has to be provided. Various types of concrete, lead, lead glass, steel and depleted uranium can be used
as shielding materials.
Thickness of the shielding must be calculated on the basis of the type, energy and activity of
radiation to keep doses received by the operators within the internationally accepted limits.
6

FIG. 6. Rear side of a hot cell with maintenance door
(Source: IPEN, Brazil).
The lead shield is equipped with viewing windows to overlook the area of operations in the
cells. A 5 cm thick lead wall requires 10 cm thick windows of standard density of 5.2 g/cm
3
. A lead
shield is easily made of bricks with antimony content of 4-7% to increase the mechanical strength. The
windows are mounted in frames fitting to the lead wall. To obtain the most economical result,
shielding walls are to be placed as close to the source of radiation as is practically possible.
Remote-handling tongs and manipulators of various types may be obtained from commercial
suppliers. Remote Handling Tongs with detachable heads allow change of the types of jaws without
removing the tongs from the box. Sphere joints are used for tong handling through the lead walls. The
whole joint is put together as a single removable unit.
Prior to putting a newly constructed hot cell into operation, a dose map indicating potential
leakage of the radiation is to be drawn up using a sealed source containing similar isotopes as intended
to be handled inside the hot cell and having known radioactivity.
As design considerations and dose calculations are well established principles applied for a long
time, no dramatic changes in hot cell design from radioactivity and contamination point of view on
conventional hot cells have been introduced. Some important considerations for the design and
construction of the heavily shielded conventional hot cells to be used for extremely high activities are
discussed below.
Adequate shielding
Optimization of radiation protection and safety principles need to be applied, with provision for
accident conditions where the source moves to the minimum distance from the operator, while
designing the shielding. Possible increase in future operations is to be considered at the time of
7

planning. Lead, and to a lesser extent steel, are the main shielding materials used for the smaller cells
while concrete, both high and standard density, is popular for large cells. Although liquid filled
windows are still in use, lead glass is preferred because of the perceived safety after an accident.
Access
The introduction of targets and reagents, and removal of product and waste require transfer
systems that are reliable and safe. The safety encompasses that of radiation, contamination and
conventional. There are a number of systems commercially available, such as the Padirac system from
the CEA, which can be used for both input and output, but due consideration should be given to the
exact requirements of the facility. An advantage of the Padirac and its inner container is the capability
of introduction of sterilized items into the cell. The inner container can be coupled to a dedicated
autoclave or similar equipment where the container and contents are sterilized and sealed. Liquid
waste can be handled through shielded drains or absorbed on some suitable material and treated as
solid. Although the latter appears to be inefficient, it may be advantageous when small volumes are
produced. The drain method may require expensive maintenance.
Manipulation
There are two basic designs for handling equipment, the tong and master-slave manipulator. The
tong, which could be seen as a fixed or manoeuvrable rod (with two degrees of freedom) is usually
limited to shielding walls up to 150 mm thick as the lead ball, which gives it the swivel action,
becomes too heavy in thicker walls.
The master slave manipulator provides far greater articulation but requires greater maintenance
and is more costly (Fig. 7). Both tools require booting to effect air tightness to the box and the
bootings should be easily replaceable.
FIG. 7. Highly shielded hot cells with master slave manipulators
(Source: NECSA, South Africa).
8
Ventilation
The box needs to be held at a controlled negative pressure, the magnitude of which is dependant
on the radio-toxicity and vapor pressure of the handled radioisotopes. Pressures of –200Pa to –500 Pa
are normally chosen. Both inlet and outlet HEPA filters are required; the inlet determines the rate of
air drawn into the box and the outlet filter must be able to handle the air flow when an opening occurs
in the box. The rate of change of air through the box is determined by the minimum requirement of the
air controller, the requirements for heat removal or the high flows that ensures aseptic clean operation
environment. For exhausting, generally 0.5 m/s is adopted as the minimum linear inward air velocity
through openings in a process enclosure used for treatment of radioactive materials.
Major changes in design will be needed to provide aseptic conditions for the production and
testing of pharmaceutical grade radioactive products.
HOT CELLS USED UNDER ASEPTIC CONDITIONS
The hot cells constructed for the production of radioactive pharmaceuticals need to meet the
requirements for a negative pressure isolator. The hot cells should be tight fitting according to the
international technical standard. The walls of the hot cells should be smooth, impervious and unbroken
and the corners are curved. Permanent installation of components, which cannot be sufficiently
cleaned, should be avoided.
Stainless steel and organic glass are recommended as construction materials. The stainless steel
surface inside the hot cell should be polished. The hot cells need to meet the general recommendations
for rooms according to the GMP regulation.
During their operation, the hot cells are under negative pressure with a 20- fold air change per
hour in case of handling radioactive pharmaceuticals. The in-air and exhausted air should pass through
HEPA filters. Airflow should be controlled inside the hot cell. The sucked in air should be filtered.
The hot cell can be equipped with double door air locks. It is desirable to provide for a
connection with a commercially available disinfector (e.g. hydrogen peroxide) and to have the front
and/or back wall vertically movable in order to clean or change the devices inside the hot cell (Fig. 8).
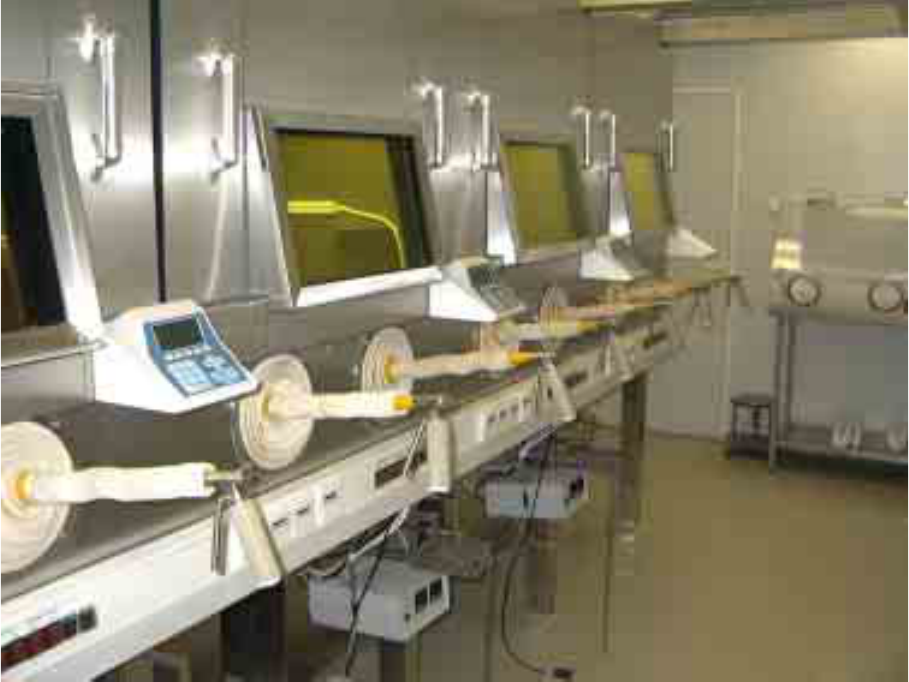
The so-called Mini-cells are commercially offered especially for the PET-technology (Fig. 9).
The Mini-cells are used for the automatic synthesis of PET pharmaceuticals, the dispensing of
pharmaceuticals in vials and syringes. The Mini-cell is a completely closed hot cell with controlled air
flow (clean room class C and A are possible, refer Table 1). The Mini-cell is designed without
manipulators.
9

FIG. 8. Hot cell with openable rear wall
(Source:Wälischmiller GmbH, Germany).
10

FIG. 9. Mini cell for dispensing with controlled air flow
(Source:Wälischmiller GmbH, Germany).
MANIPULATORS USED FOR HOT CELLS
Different manipulators are used for the handling of radioactive materials inside the hot cells
(negative pressure isolator) for the production of radioactive pharmaceuticals. The manipulators have
to be installed absolutely tightly. The use of ball tong manipulators and master slave manipulators
depends on the necessary thickness of the shielding wall and the necessary manipulations inside the
hot cell. Ball tong manipulators can be used up to a shielding thickness of 100 to 150 mm lead.
Master slave manipulators are to be used if the shielding thickness is greater than 100 to 150
mm lead. The master slave manipulators are recommended if complicated and sensitive operations
inside the hot cell have to be carried out (Figs 10–11). The booting of the manipulators are to be
resistant to agents, which is used for cleaning and disinfecting as well as the chemicals used in the
production.
11
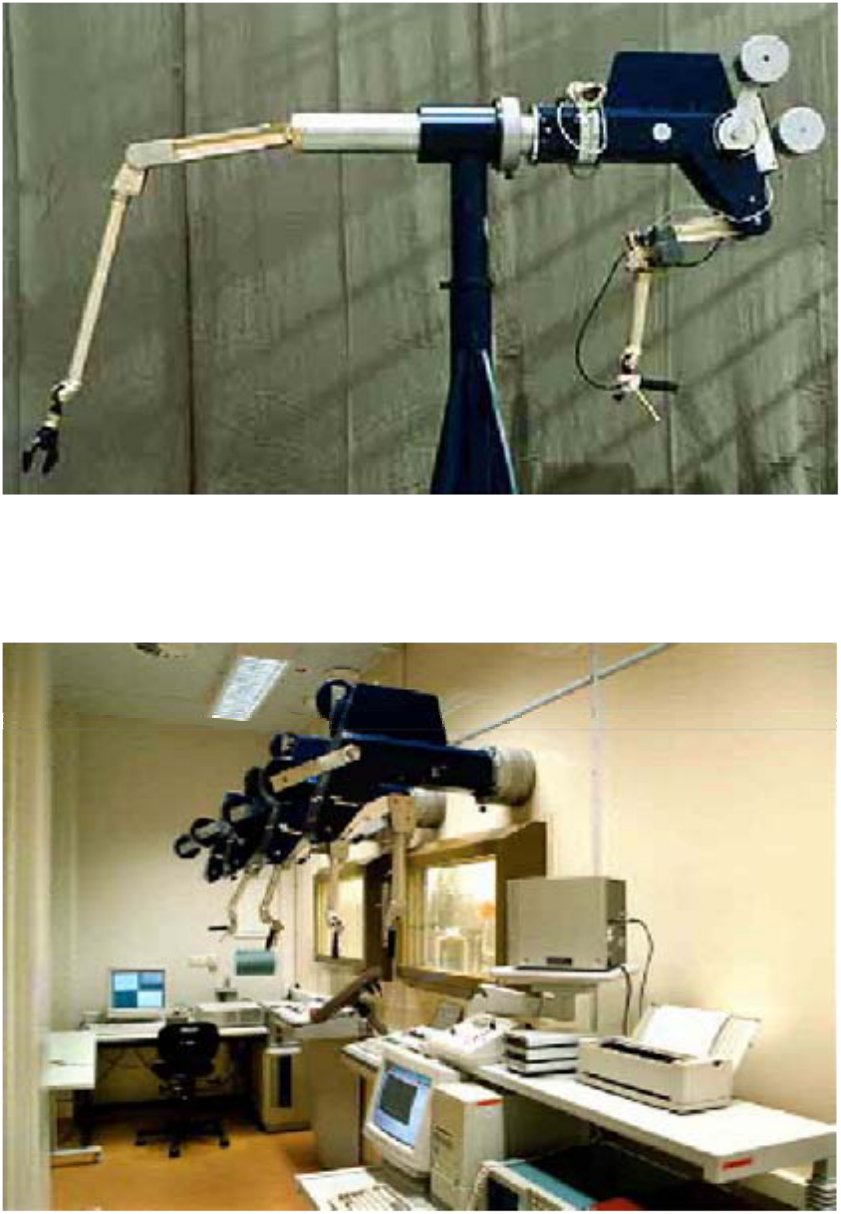
FIG. 10. Master slave manipulator for total remote handling
(Source:Wälischmiller GmbH, Germany).
FIG. 11. Hot cell with installed master slave manipulators
(Source: Wälischmiller GmbH, Germany).
12

MAINTENANCE OF HOT CELLS, LEAK TESTING FOR ASEPTIC HOT CELLS
The maintenance of the cell and in-cell equipment is technically demanding due to the high
radiation and contamination levels experienced when the integrity of the facility is broken. This
problem is to be addressed in the design stage and all routine maintenance operations are to be
practiced in the commissioning stages. The routine service such as lubrication of joints and the
adjustment of manipulator cables or tapes are examples of operations that do not pose undue
radiological risk, whereas the replacement of booting does pose radiation hazard.
The standard manipulator access through the biological shield is located at the top of the cell
allowing the safe removal of the manipulator. The replacement of its booting should be an easy
operation subsequent to that. The removal of the tong and ball, required for its booting replacement,
exposes the personnel to the cell’s radiation. This implies that consideration be given to the
development of procedures and designs to avoid this possibly irretrievable situation. Some of the
useful devices are given in Figs 12–13.
Cell doors and port closures should be made to allow replacement, and cell lighting should be
protected and placed outside the containment box if possible.
The outlet filters can become a radiation and contamination risk. It is recognized in the latest
designs that these filters should be removed through the cell’s waste system, as would booting and
defective slave-hands.
In-cell equipment is usually produced or adapted in-house and therefore does not generally
exhibit the same level of design refinement as commercially available cell equipment. Nevertheless,
consideration should be given to factors such as little or no maintenance, the possibility of strategic
component in-cell repair or replacement the necessity for cleaning and the avoidance of superfluous
extras.
FIG. 12. Tong manipulator with accessories
(Source: Wälischmiller GmbH, Germany).
13

FIG. 13. Ball brick for the installation of the tong manipulator
(Source: Wälischmiller GmbH, Germany).
While conventional hot cells need only periodic decontamination, aseptic hot cells need
sanitation too. Neither generally settled solution nor good applicator recommendable for this purpose
are readily available. In most cases working surfaces are cleaned regularly by means of manipulators
and full cleaning is possible only during maintenance periods. For sanitizing aseptic hot cells designed
as isolator or operated in clean room, various spraying and evaporation methods are available.
Sanitation of aseptic hot cells can be carried out by any of the following methods:
ʊ spray method with H
2
O
2
solution
ʊ evaporation of H
2
O
2
ʊ combination of saturated water steam/H
2
O
2
ʊ flowing saturated water steam
ʊ different peracetic acid procedures
ʊ formalin methods, etc.
14

To achieve clean grade, equipment and devices generating particles and/or vapours, aseptic hot
cells (driving motors, vacuum pumps, cranes) - as far as possible - should be placed and operated
outside the hot cell. Machinery of freeze-dryers should also be placed outside the clean room, only
chamber should be opened to the clean area.
3.
ASEPTIC CONDITIONS IN THE PRODUCTION OF
RADIOPHARMACEUTICALS
Medicinal or medical products should be protected from microbiological contamination by their
environment. As the direction of the potential microbiological contamination is from the environment
and towards the material the ideal protection is to hinder movement of the surrounding air towards the
product. This can be achieved by providing filtered airflow towards the product from the environment.
Basically such a system creates overpressure in the production area so clean rooms operate with
positive pressure relative to the environment.
According to the requirements of the GMP relating to medicinal products, sterile
pharmaceuticals should be manufactured in clean areas, entry to which should be through airlocks for
personnel and/or for equipment and materials. Clean areas should be maintained to an appropriate
cleanliness standard and supplied with air, which has passed through filters of an appropriate
efficiency. This protects the product from microbiological contamination by the environment.
Categories of clean areas according to the number of suspending particles and that of
microorganisms in the air are given in Table I.
TABLE I. CATEGORIES OF CLEAN AREAS
CLEAN GRADE Max. number of
Particles equal or above
0.5 µm
Max. number of particles
equal or above 5 µm
Maximum number of
Microorganisms
A
(3 500 / m
3
)
(100 / ft
3)
0 / m
3
(0 / ft
3
)
0 / m
3
(0 / ft
3
)
B
3 500 / m
3
(100 / ft
3
)
0 / m
3
(0 / ft
3
)
5 / m
3
(<1 / ft
3)
C
350 000 / m
3
(10 000 / ft
3)
2 100 / m
3
(60 / ft
3)
100 / m
3
(3 / ft
3)
D
3 500 000 / m
3
(100 000 / ft
3)
21 000 / m
3
(600 / ft
3)
500 / m
3
(14 / ft
3)
Clean areas provide aseptic conditions for the manufacture of medicinal products for which
basically the number of suspended particles (which carry bacteria) in the air should be reduced and at
the same time working surfaces should be sanitized and microbiological contamination from other
sources should be reduced (e.g. by protecting clothing).
For creating aseptic processing conditions two technical solutions have been developed which
are discussed below:
15
CLEAN ROOMS
Clean rooms are laboratories or plant-sized clean areas in which HEPA filtered air is
continuously introduced and recirculated. Positive air pressure relative to the surroundings is
maintained with this air control. Personal and material flow is controlled through air locks to avoid the
change of air between the zones.
In addition to the design and construction considerations assisting effective sanitation and
limiting the spread of bacteria, measures and working rules are enforced to reduce the risk of
microbiological contamination.
ISOLATORS
These are small-sized, local clean areas, which are hermetically sealed from the environment,
excluding not only the surroundings but also the operator from the clean area. With their small sizes,
isolators provide also economical result by isolating as close to the product as practically possible.
Isolators can be supplied with doors or with air locks for supply of materials. Positive pressure
isolators and negative pressure isolators are available according to the type of air flow control within
the isolator.
For radioactive materials, the direction of potential contamination (and accordingly protection)
is the opposite to that of microbiological contamination. Direction of contamination is from the
material to the environment and for that reason the environment must be protected from vapours
and/or particles of radioactive materials. From the ventilation point of view, the air needs to be
exhausted from the shielded hot cells to maintain negative pressure within the hot cells.
As the direction of microbiological and radioactive contamination is opposite to each other, the
protection methods should be combined for producing radioactive pharmaceuticals. In addition to the
reduction of radioactive and microbiological contamination, radiation protection must also be taken
into consideration. Harmonizing the three requirements is not easy and in some cases even conflicting.
Where the conflict cannot be resolved radiation safety must have priority.
GMP guidelines are elaborated primarily for conventional pharmaceutical products and
production conditions. They do not deal with radioactive materials in the required detail giving the
basis for designing and operating radioactive facilities under aseptic conditions. The GMP describes
only specific features of radioactive materials and refers to the production conditions of sterile
pharmaceutical products. There is need to document harmonized design and operation principles
relating to radioactive pharmaceuticals in every detail (not only referring to conventional sterile
pharmaceuticals) because general solutions cannot be applied automatically for radioactive materials.
Specifications for radioactive materials are specific in the following four aspects:
To minimize the radiation dose to persons (radiation protection is necessary)
Radioactive contamination caused by the material should be eliminated
Mass of the active ingredient is extremely low so radioactive pharmaceuticals do not cause
toxicity
As administered volumes are low, dose of pyrogen materials injected is also low.
While the first two aspects are drawbacks causing significant difficulties in handling and in
combining technical conditions with GMP requirements, the latter two features are advantageous from
GMP point of view allowing reasonable compromises.
16
Harmonized solutions for the aseptic processing of radioactive pharmaceuticals are:
ʊ Placing conventional hot cells (with –ǻP) in clean rooms (with +ǻP) where exhausted hot cells
are supplied with filtered air from the surrounding clean room.
ʊ Using hot cells with own air flow control (supplied with filtered in/air and exhaust air systems),
designed and operated as negative pressure isolators (–ǻP). Such hot cells must be airtight to
avoid air sucking from the surrounding (Fig. 14).
FIG. 14. Hot cells (–ǻP) in clean room (+ǻP)
(Source: IZOTOP, Hungary).
Negative pressure isolators combine both types of protection (protection of the pharmaceutical
product from the microbiological contamination of the surroundings and protection of the
surroundings from the vapours and particles of the radioactive materials) within one space. If negative
pressure isolators are supplied with appropriate shielding against radiation, radiation protection can
also be provided. This solution is less expensive than placing conventional hot cells into high grade
clean rooms, especially for small and medium scale production capacities.
Application of hot cells designed and operated as negative pressure isolator follow the same
principle given for the hot cell design that protection is the cheapest if containment is closest to the
contamination source.
Hot cells with controlled air flow operated as negative pressure isolator (supplied with filtered
in/air and exhaust air systems) with grade A or C depending on the type of product are recommended
for radioactive materials similar to cytotoxic products in the conventional pharmaceutical industry.
The negative pressure isolator principle is well applicable also for radioactive preparation laboratories
17
in hospitals. Owing to the harmful character of radioactive materials air extracted from hot cells
should not be recycled.
Based on the above considerations, when processing radioactive materials under aseptic
conditions for use as pharmaceuticals, the following criteria can be adopted.
ʊ For terminally not sterilized products (typically Tc-kits), ‘A’ class clean area with ‘B’ class
background should be provided.
ʊ For terminally sterilized products (e.g. autoclaved or filtered radioactive solutions) ‘C’ class
clean area is recommended. Background class depends on the following considerations:
ʊ If conventional hot cells are placed in clean room (air is sucked into the hot cell from the
background) a clean room class dictated by the product treatment (selected according to the
above principles) should be provided.
ʊ If hot cells are self-supplied with controlled and filtered air (designed and operated on the
negative pressure isolator principle) the inside clean class should be selected according to the
product treatment (see above principles) and ‘D’ class clean room as background is
recommended.
As design principles of ‘D’ class clean rooms are very close to those of radioisotope laboratories
and as radioisotope laboratories are already equipped with ventilation, zoning system and changing
rooms, a ‘D’ class clean room can be created and operated without serious additional investments, by
replacing the existing low-grade filters to bacteria retardant HEPA filters; by supplementing cleaning
agents with disinfection agents and by applying simple personal protection (changing aprons and
covering hair).
As radioisotope handling facilities are generally big halls equipped with heavy machinery (e.g.
cranes for lifting and transporting containers) which generate particles, and because the target
treatment includes several dirty mechanical operations (e.g. target crushing, cutting) as well as several
target processing technologies requiring operations with acid addition and evaporation, powder
treatment, high temperatures, etc. these operations should be separated from further processing (e.g.
bulk dilution, adjustment of radioactive concentration, dispensing, autoclaving, packaging) to allow
aseptic conditions to prevail.
Because the final product (radioactive pharmaceutical) will be terminally sterilized (accordingly
eliminating its bacteria and fungi contamination) and because the result of the target processing is a
concentrated bulk solution which will be considerably diluted in the next phase of processing
(accordingly ensuring that its pyrogen concentration is considerably lowered below the acceptable
limit), acceptable microbiological purity can be expected from such separation of the technological
operations (which must be validated).
However, filtering the bulk solution and using pyrogen-free distilled water throughout the whole
technology, including dilution is recommended. In a typical dilution rate of 100x the majority of the
product consists of sterile and pyrogen-free distilled water, which will determine its microbiological
character.
With such a separation conventional hot cells can be used for target processing and hot cells
operated under aseptic conditions should be used for pharmaceutical operations. The same separation
can be applied to situations where radioactive bulk is imported and processed to pharmaceutical grade
radioactive products. A drug master file characterizing the bulk and the radioactive pharmaceuticals
and describing the procedure and conditions of the preparation is necessary in order to comply with
GMP requirements.
Lead containers serving as shielding and providing secondary packaging material represent high
mass and volume. To avoid introduction of lead containers into clean areas, it is recommended that
well designed air locks be connected to the background or to the outside area. In this way, only
18
ampoules should be entered and treated in the inside clean area as is the case in conventional
pharmaceutical plants. This is recommended because heavy lead containers are difficult to disinfect
and can emit particles contaminating the clean area.
For disinfecting inside surfaces an H
2
O
2
generator is the most sophisticated and most effective
tool. Spraying disinfection agents and/or alcohol with a concentration of 70% is also applied. The
evaporation and circulation method of H
2
O
2
- solution proves successful as a method with a good
validation possibility. Suitable automatic devices on the basis of this H
2
O
2
- evaporation method are
commercially available. Spraying bottles containing isopropyl-alcohol are commercially available and
other known disinfectants can also be used by spraying. Whatever type of cleaning and sanitation
method is selected, its efficiency and suitability must to be validated.
Cleaning and sanitation are important preparations to provide aseptic conditions for production.
However, hot cells generally cannot be opened regularly for cleaning. Although, effective sanitation
agents and decontamination agents are available, there is no easy-to-use applicator, unless hydrogen
peroxide spraying head is installed. In the absence of hydrogen peroxide generator spraying for
sanitizing agents, isopropyl or ethyl alcohol may be generally applied onto the surfaces. Efficiency of
cleaning and sanitation methods needs to be validated.
Authorities in the Republic of Korea require adherence to GMP requirements for air quality in
production of radioactive pharmaceuticals. In spite of significant improvements of hardware related to
GMP, practically there are many conflicts in implementation of this regulation for production of
radiopharmaceuticals due to the differences between radioisotopes and pharmaceuticals. In principle,
it would be solved economically through the harmonisation of hardware and software related to GMP
requirements within an achievable range. GMP requirements can be met by having good systems
based on equipment and facilities as well as validation based on software. Conflicts occurring from the
different properties between radioisotopes and pharmaceuticals need to be solved by a radioisotope
producer by reflection of special circumstances due to radiation.
In the Republic of Korea, the committee of Korea GMP (KGMP) estimation recommended that
the GMP guideline for production of radioactive pharmaceuticals be adopted. In principle, regulations
for the general pharmaceuticals are correspondingly applied to radioactive pharmaceuticals, but the
GMP regulations are not specific to radioactive pharmaceuticals. The production facilities for
radioactive pharmaceuticals were completed in accordance with KGMP in 1998. As the regulation
authority encourages adopting the regulation of KGMP certification, documents for the application of
GMP certification are being prepared. In future all items such as medical radioisotopes, cold kit and
radiation therapy sources need to be controlled by the code and standards of the Korean Drugs,
Cosmetics and Medical Instruments under the KGMP regulation.
Recently the dose limit was drastically reduced from 50 to 20 mSv/man/y. In order to reduce the
man/mSv exposition during the radiation work, it is essential to develop suitable accessories/ devices.
Despite all the above difficulties, radioisotope production must continue economically. It has
been said that the first responsibility of a radioisotope producer is to define reality. The
radiopharmaceutical production facilities are trying not only to survive these tough regulatory norms
but also want to grow in these circumstances and leading the way by supporting health care programs
in the world.
In order to solve these problems it is worthwhile to take into account the technological advances
made in the past few decades especially in the field of remote handling and robotics. These measures
will also reduce the human intervention during the production and quality control of radioisotopes. It
is therefore essential to adopt automated operations for radioisotope production in future.
19
Parameters and methods applied for quality control of aseptic areas are as follows:
ʊ Measuring the number of particles in the air with a particle counter e.g. Biotest APC Plus
particle counter;
ʊ Determining air microbiological contamination: e.g. Biotest RCS Plus air sampler with medium;
ʊ Determining bacterial contamination of the product: e.g. culture on thyoglicolate-Bouillon
medium;
ʊ Determining fungal contamination of the product: e.g. culture on Caso/Bouillon medium;
ʊ Determining pyrogen concentration in the product: e.g. LAL test.
Prior to operating aseptic areas (hot cells/isolators) they need to be qualified for the following
parameters:
ʊ Determining number of particles in air
ʊ Determining microbiological contamination in air
ʊ Measuring pressure, relative humidity and temperature
ʊ Determining air flow rate
ʊ Determining air exchange rate
ʊ Determining air flow velocity
ʊ Checking integrity of the HEPA filters
ʊ Leak testing
ʊ Determining cleaning rate of air.
In addition to equipment qualification, process validation is also required by quality assurance
relating to pharmaceuticals (GMP). Critical parameters of the processes are to be incorporated into
validation.
4.
EQUIPMENT AND DEVICES FOR USE IN HOT CELLS
Design considerations for devices and equipment are as follows:
ʊ Compact in size
ʊ Operable with manipulator
ʊ Drives and control devices to be placed outside the hot cell and
ʊ Easy maintenance.
Devices presented by participants included dispenser, autoclave, peristaltic pumps,
capping/decapping tools, target crusher, target cutter, container opening devices, can opener, activity
transport lift system, pneumatic or electrical crane operation. A brief discussion on these is given
below.
TARGET OPENER
Consists of two cylinders rotated by an electric motor and a cutting disc (Fig. 15). Aluminium
capsule is laid on the cylinders and the cutting disc is pushed to the aluminium capsule while rotating.
20

FIG. 15. Target opener
(Source: IZOTOP, Hungary).
AMPOULE CAPPING DEVICE
It is a pneumatically operated crimper (Fig. 16). The ampoule with its cap is placed on its holder
by manipulator and then the pneumatic sealing head is operated. While doing the operation in clean
room, filtered air need to be introduced for pneumatic operations and the exhaust air has to be left
outside the clean area.
21

FIG. 16. Ampoule capping device
(Source: IZOTOP, Hungary).
AUTOCLAVE
Small sized, manipulator-operated sterilization chamber with two trays for vials serves as
autoclave (Fig. 17). Temperature and pressure controlled. Autoclaving is executed automatically by
switching the control instrument on and is thereafter controlled for the main parameters (temperature,
pressure, time). Measured parameters are displayed and recorded. Qualification of the equipment
includes parameter verification, temperature distribution and sterilisation efficiency verified by test
bacteria.
22

FIG. 17. Compact sized autoclave for ampoules
(Source: IZOTOP, Hungary).
Pass-through autoclave
This is built into the hot cell or between two hot cells as an airlock providing transfer of the
autoclaved product from the hot cell to the package area or from one hot cell to another (Fig. 18).
FIG. 18. Autoclave built in the hot cell
(Source: IPEN, Brazil).
23

PERISTALTIC PUMP OPERATED WITH MANIPULATOR
Suitably modified peristaltic pump can be of great use in side the hot cell. A short arm serves to
press the head of the pump down and to lift it up by means of manipulators (Fig. 19). Tubes can also
be placed into the pump by means of manipulators. Such peristaltic pumps are commercially available.
FIG. 19. Peristaltic pump operated with manipulator
(Source: Commercially available).
ACTIVITY TRANSPORT LIFT SYSTEM
The activity meter has to be tightly installed under the hot cell. The measuring chamber is a part
of the hot cell and covered by a lid in the rest state (Fig. 20). It is recommended that a plastic insert be
used inside the chamber, which can be easily removed and cleaned. The sample to be measured in the
chamber is moved by an electrically controlled, vertical lifting device.
24

FIG. 20. Transport lift system for sample radioactivity measurement
(Source: Wälischmiller GmbH, Germany).
DRY DISTILLATION EQUIPMENT FOR THE PRODUCTION OF
125
I
This consists of a compact electric oven for heating up the irradiated aluminium capsule to
release the desorbed iodine, an acid scrubber and alkaline absorbers (Fig. 21). The oven is temperature
controlled and values are displayed.
FIG. 21. Dry distillation equipment for
125
I production
(Source: IZOTOP, Hungary).
25

DRY DISTILLATION EQUIPMENT FOR THE PRODUCTION OF
131
I
This consists of a compact electric oven for heating up the irradiated tellurium oxide granules
contained in a silicium-carbide ceramic pot, followed by an acidic scrubber and alkaline absorbers
(Fig. 22). Temperature controlled and supplied with a built-in activity meter.
FIG. 22. Dry distillation equipment for
131
I production
(Source: IZOTOP, Hungary).
PRODUCT SPECIFIC EQUIPMENT
These in cell equipment serve in the production of specific radioactive pharmaceuticals. The
equipmnet for production of
131
I capsules is used for filling adsorbent in the first step followed by
dispensing iodine solution and then capping the capsule (Fig. 23). Filled up capsules are dropped into
lead container. Other examples are technological equipment for
67
Ga (Fig. 24) and
131
I-mIBG (Fig. 25)
production.
26

FIG. 23.
131
I capsule filling equipment
(Source: KAERI, Republic of Korea).
FIG. 24. Technological equipment for
67
Ga production
(Source: IPEN, Brazil).
27

FIG. 25. Technological equipment for
131
I-mIBG production.
(Source: IPEN, Brazil)
5.
AUTOMATION, MECHANIZATION AND PC CONTROLLED PROCESSING
In the production of radioactive materials automation is important for the following reasons:
ʊ Safety (reduces dose to personnel)
ʊ Improved quality of product (e.g. welding seam)
ʊ Speeds up production
ʊ Ideal for large number of repetitive high dose and complex operations
ʊ More GMP friendly – for automatic data records for GMP
ʊ Fields which typically have been developed – PET isotopes, sealed sources,
201
Tl and
67
Ga,
131
I
capsules,
99m
Tc solvent extraction
ʊ Allows further automation /mechanisation where advised
ʊ Cost/benefit combination.
There are many r
e
e
a
a
s
s
o
o
n
n
s
s
f
f
o
o
r
r
a
a
u
u
t
t
o
o
m
m
a
a
t
t
i
i
o
o
n
n
:
:
ʊ To reduce personnel exposure to radiation
ʊ To prevent human error for reliable quality control and precise work
ʊ For mass production to improve the production rate
ʊ To easily maintain the clean class without the human intervention
ʊ To solve problems related to processing time, space and hazard.
28

Fig. 26 shows a typical automation system in sealed sources production in the Republic of
Korea.
FIG. 26. Automation in sealed sources production
(Source: KAERI, Republic of Korea).
D
D
u
u
r
r
i
i
n
n
g
g
t
t
h
h
e
e
p
p
l
l
a
a
n
n
n
n
i
i
n
n
g
g
s
s
t
t
a
a
g
g
e
e
o
o
f
f
a
a
u
u
t
t
o
o
m
m
a
a
t
t
i
i
o
o
n
n
p
p
r
r
o
o
c
c
e
e
s
s
s
s
,
,
i
i
t
t
i
i
s
s
i
i
d
d
e
e
a
a
l
l
t
t
o
o
p
p
e
e
r
r
f
f
o
o
r
r
m
m
a
a
f
feasibility analysis,
proof-of-concept, benchmarking and prototyping for the process. In the development stage, concept
design including software and detailed drawings are to be carried out. In the final stage, manufacture
and installation of the mechanical parts and system operation and integration are done. The automatic
processing system for
18
F-FDG synthesis modules was introduced in PET centers. At present synthesis
module for production of SPECT radioactive pharmaceuticals such as m
123
IBG synthesizer are
available.
In case of m
123
IBG module, the installation of a manual apparatus for the chemical process will
need bigger space for installation and hence it is difficult to maintain clean class due to human
intervention. In the field of PET radioactive pharmaceuticals, synthesis modules are very useful for
saving the space and time as well as assuring the reliability in quality and routine supply. At present,
the process automation in production of radioisotopes is expected to show many advantages but the
introduction of full automation system for commercial supply will be considered on the basis of the
investment for system development, maintenance, radiation effects of electric sensors, reliability of
system operation, etc. In future it is hoped to have commercial standard component supplied as a
package for automation of each process.
There are two kinds of machine in
131
I capsule manufacture used in Republic of Korea (Fig. 27).
One is for therapeutic and the other is for diagnostic capsules. The main problems are solution
dispensing and assembling the upper and lower capsules. The use of disposable syringe can be
considered for economical and convenient preparation of diagnostic capsules. Dispensing precision is
very important because small amounts of highly concentrated
131
I solution are used for production of
therapeutic capsules. It is recommended that automatic systems be partially introduced for solution
dispensing in order to improve reliability of dispensing precision and productivity.
29

FIG. 27. Automated
131
I therapeutic capsule filling system
(Source: KAERI, Republic of Korea).
Further examples for automation in radioisotope production are found throughout this document
under specific devices and/or products.
PC controlled processing not only provides high-level automation and reliability for the
operations but also GMP conforming data registration and automatic document printouts.
6.
EXAMPLES OF MAJOR RADIOISOTOPE PRODUCTION SYSTEMS
99
Mo-
99m
Tc GENERATOR PRODUCTION LINES
Due to its favourable nuclear and chemical properties
99m
Tc is the most frequently used
radionuclide worldwide in nuclear medicine for making millions of diagnostic images. It is obtained
from three basically different sources:
ʊ Eluted from fission
99
Mo based chromatographic column generator
ʊ Eluted from irradiated
99
Mo based gel-generator
ʊ Separated from irradiated
99
Mo compound by solvent-extraction.
Fission
99
Mo based chromatographic
99
Mo-
99m
Tc generators became generally used in practice
due to the simple processing and handling, reliability of product quality and high radioactive
concentration. At the same time considerable efforts have been made to develop
99
Mo neutron
irradiation based on so called ‘alternative generators’ with acceptable radioactive concentration.
30
Production centers developed different technical solutions for different scales of manufacture
that are described below:
Ampoule filling batch method based on fission
99
Mo
This is used to produce small number of fission
99
Mo based generators. The procedure for the
production of the generators is divided into three phases: the inactive preparation and assembling, the
loading process and the final packaging. The aseptic assembling of the generators can be carried out
completely as an inactive procedure. In this case an inactive test of the complete assembled generator
system is possible.
The loading procedure of the generators, (handling the radioactive material), need to be as
simple as possible. The loading steps are:
ʊ dispensing the necessary portion of loading solution in vials
ʊ steam-sterilisation of vials
ʊ loading procedure according to the elution procedure.
The realization of the loading process according to the GMP-requirements can be carried out
remotely controlled under a laminar flow module into a negative pressure isolator (shielded hot cell).
The loading procedure can be carried out with an automatic system including sterile filtration. The
packaging procedure need be automated as much as possible in order to reduce radiation exposure to
staff.
Automated or semi-automated
99
Mo dispensing system and Tc-generator production line
Most production methods for
99m
Tc generators follow the steps of stock solution preparation, the
loading of the Al
2
0
3
column with measured aliquot of the solution, the possible sterilization of the
column, the assembly of the column in a lead pot with its elution needles and finally the complete
assembly of the box or container. A degree of quality control testing usually precedes the packaging
for transport.
Large scale generator production has necessitated the automation of some or all of the above
steps. This is mainly due to reasons of quality in the first half and radiation safety in the second half of
the process. A modern innovation is the loading of the generator column already placed in the lead pot
and assembly. This implies a facility constructed from a standard cell coupled to an aseptic shielded
assembly line. The active liquid is accurately measured and pumped through the wall of the cell
directly into the pre-assembled generator in the aseptic area. Removable shields protect the operator
while loading is taking place. A conveyor system takes the generator assembly to the packaging and
dispatch areas.
The above system serve for producing large numbers of generators. Automatic dispensing
techniques have also been developed, whereby dispensing takes place by vacuum suction of the
dispensed
99
Mo solution through the chromatographic column. Column sterilisation is made before or
after dispensing.
Dry
99
Mo bulk intended for supply in the near future by NECSA will provide low rate radiolysis
with no liquid. If sterile solvent is used for dissolving the dry
99
Mo, extremely low pyrogenity of the
product will be achieved by diluting the bulk with sterile, pyrogen-free distilled water or saline.
As Tc-generators produced from fission
99
Mo contain a built-in bacteria filter for the eluate,
such generators are considered as terminally sterilized products needing ‘C’ class clean room for
processing. However, in places where injection needles are connected to the column, microbiological
31

contamination should be considered as critical and hence local A - grade air flow or such grade hot
cell is recommended for the critical operations.
GEL GENERATORS
In the production of
99m
Tc generators the gel generator option based on reactor irradiated
molybdenum (i.e. low specific activity) provides an alternative to the fission generators. The irradiated
molybdenum is incorporated into a zirconium molybdate gel, which is eluted with saline in the same
way as fission generators representing identical qualities.
The gel generator utilizes low specific activity (n,Ȗ) produced
99
Mo which is processed post-
irradiation into an insoluble zirconium molybdate hydrous gel structure. The dried gel contains about
25% by weight of molybdenum and has properties consistent with a cation exchanger.
The gel is insoluble and chemically stable within the pH range 2-9. It successfully withstands
thermal (wet steam) autoclaving and consequently the gel generator may be presented as a terminally
sterilized product
The passage of an aqueous eluant (typically either pure water or physiological saline) through
the column of the gel releases the
99m
TcO
4
-
. The chromatographic separation of
99m
TcO
4
-
from the gel
column can be performed with the same degree of ease like the fission
99
Mo generator (Fig. 28).
FIG. 28. Internal view of
99
Mo-
99m
Tc gel generator hot cell
(Source: IPEN, Brazil).
32

The external view of a
99
Mo-
99m
Tc column generator production facility is shown in Fig. 29.
FIG. 29.
99
Mo-
99m
Tc generator production line
(Source: IPEN, Brazil).
CYCLOTRON AND
18
F-FDG SYNTHESIS MODULES
Several radioisotopes for medical use can be produced in cyclotron by charged particle (protons,
deuterons or alphas) irradiation. These include isotopes such as
201
Tl,
67
Ga,
111
In,
123
I,
81
Rb,
18
F,
13
N,
11
C and
15
O. The last four isotopes are used for positron emission tomography (PET) studies.
Cyclotrons with energy in a range of 10–70 MeV for proton irradiation and of 5–20 MeV for deuteron
irradiation are commercially available and the manufacturers offer cyclotron suitable for the planned
application (Fig. 30).
33

FIG. 30. Cyclotrons with different proton energy
(Source: KAERI, Republic of Korea).
Radioisotope production in Cyclotrons in Republic of Korea:
ʊ MC-50 Cyclotron (50MeV proton, installed in 1986)
ʊ KIRAMS-13 (13 MeV, installed in 2002)
ʊ Cyclone 30 (30 MeV, under installation).
Among the cyclotron produced radioisotopes,
18
F is most widely used due to the general
occurrence of glucose metabolism in biological systems. The
18
F-FDG synthesis can be run on
commercially available automated synthesis modules.
18
F-FDG synthesis panels are the best
automated systems among radioisotope technologies. Installation according to the instructions of the
supplier is the only requirement in most cases.
18
F-FDG synthesis consists of the following steps:
ʊ Separation of
18
F from target by ion-exchange
ʊ Introduction of
18
F into organic phase with KF through phase transfer catalyst
ʊ Fluorination of FDG precursor (mannose-triflate) by nucleophylic substitution
ʊ Hydrolysis of the protecting groups by acid or alkali addition
ʊ Separation of
18
F-FDG from reaction mixture through series of separation cartridges.
Typical synthesis time is less than 30 min providing a radiochemical yield around 70% and
radiochemical purity of >99%. Residual activity on the panel is typically <1%.
For the routine production of
18
F-FDG a radiochemistry laboratory with a lead shielded hot cell
for safe processing and easy, available maintenance should be provided. Both the hot cell and the
laboratory must comply with requirements relating to aseptic production conditions and those of GMP.
MC-50 Cyclotron Cyclone-30 KIRAMS-13
34

Cyclotron irradiation does not need an aseptic environment. FDG panel need to be placed into
class ‘A’ hot cell designed and operated isolator with D grade background (Fig. 31). In case of batch-
type (openable) hot cell. A clean grade is reached within the validated time after closing the hot cell
and switching the air flow on.
FIG. 31. FDG synthesis module
(Source: KAERI, Republic of Korea).
Dispenser
It is recommended that dispensers be used for the dispensing of radioactive pharmaceuticals
where the manipulator can easily change the dispensing tube or syringe. The changeable tube or
syringe should be available as sterilized components. The dispensing unit needs to have provision to
guarantee the complete documentation of the dispensing procedure. A dispensing unit used in IPEN,
Brazil is given, Fig. 32.
The module can be operated in class ‘C’ clean room if the filtration and dispensing is carried out
in parts in another mini cell. Class A would be required only in cases where the complete process is
carried out. For background at least class ‘D’ is recommended.
Before Installation After Installation
35

FIG. 32. FDG dispenser for use in isolator
(Source: IPEN, Brazil).
The product solution is dispensed into open ampoules, which are closed with rubber closure and
capped automatically. Air used by the built-in capping device for pneumatic operation need be
bacteria filtered. If already closed and capped ampoules are used for dispensing, leak tightness of the
ampoules need to be validated after several piercing.
Filled and closed ampoules are transferred to lead containers through an airlock operated by
valves. Lead containers (as secondary packaging materials) are introduced only into the background
clean room after spraying with 7`% alcohol.
It is very difficult to achieve effective cleaning of hot cells used for radionuclides with half life
longer than 24 hours. The isolator is designed in such a way that the inner surfaces may be cleaned
easily, have rounded corners; and doors or movable walls can be operated with ease. The necessary in-
cell equipment need be as simple as possible, easy to clean and using sterilized one way components
whereever possible. Installation of equipment generating particles and/or vapours needs to be avoided.
If inner areas of the isolator are not accessible for cleaning because the isolator cannot be opened after
each batch (for example the cleaning of a box by manipulator which is used for the handling of
radionuclides with half life longer than 24 hours) then the frequency of dismantling the isolator and
performing thorough cleaning needs justification.
A balance needs to be drawn between the frequency of complete cleaning and frequency of
dismantling the isolator. The user of the isolator has to make and document a risk assessment
justifying its practices.
36

Care has to be taken to see that the cleaning agents are compatible with the material used for the
isolator construction (e.g. stainless steel, polymeric materials etc.) and the installed in-cell equipment
(Fig. 33). Residue of cleaning agents is removed prior to sanitization in order to prevent the masking
of micro-organisms by the sanitizing agent.
FIG. 33. Clean mini cell designed as isolator for PET-module operation
(Source: Wälischmiller GmbH, Germany).
SEALED SOURCES PRODUCTION
Radioactive sealed sources are encapsulated in a suitable container or prepared in a form
providing equivalent protection against mechanical disruption. In some cases protection against heat
and corrosion effects also may be required. Production of radioactive sealed sources is carried out in
shielded conventional hot cells supplied with master/slave manipulators or tongs.
Sealed radioactive sources are widely used for industrial (
192
Ir for radiography,
60
Co for gamma
irradiators,
60
Co and
137
Cs for process control measurements and gauging) and medical (
192
Ir and
125
I
for brachytherapy,
60
Co for teletherapy) applications.
Electric arc welding is generally used for capsule sealing and laser welding for miniature
sources. Typical arc welding current is 10-20 Amp with welding speed of 4-5 mm/s at an electrode
distance of <1 mm and rotation speed of 20 per min. Welding needs high precision to provide good
quality welding seam and leak-tightness of the capsules. Leak test is generally made with nitric acid in
an ultrasonic bath. To satisfy these technical needs automated welding machines with capsule rotating
tools have been developed.
In addition to the conventional use of radioactive sealed sources, miniature size radioactive
sealed sources of
192
Ir,
125
I and
103
Pd are being increasingly used for brachytherapy of cancer.
37

The production of such sealed sources together with that of the conventional sealed sources
such as
192
Ir and
60
Co also require remote precision welding using laser or arc and microprocessor
controlled positioning devices (Figs 34–36).
FIG. 34. Welding system for
60
Co sealed source production
(Source: KAERI, Republic of Korea).
Components
Welding chamber
TIG Welder
Vacuum pump : oil
Control system : PLC
Dimension in mm;
W : 371, D : 350, H : 725
Production rate :
5ea/hr
38

FIG. 35. Welding machine for
192
Ir sealed source production
(Source:KAERI, Republic of Korea).
FIG. 36.
192
Ir sealed source for brachytherapy
(Source: KAERI, Republic of Korea).
As production of radioactive sealed sources consists of mechanical processes, the rate of
automation is the highest in this field. Procedures such as capsule loading, array, welding, welding
seam control, marking, unloading, storage, inventory, etc. are generally fully automated processes
contributing to the reliability, precision, decreased radiation dose and increased production capacity.
W
W
i
i
r
r
e
e
C
C
u
u
t
t
t
t
i
i
n
n
g
g
,
,
W
W
e
e
l
l
d
d
i
i
n
n
g
g
,
,
P
P
o
o
l
l
i
i
s
s
h
h
i
i
n
n
g
g
S
S
y
y
s
s
t
t
e
e
m
m
Ir-192 Source Capsule
Welding System
Nd-
Y
AG Lase
r
system
39
BIBLIOGRAPHY
INTERNATIONAL ATOMIC ENERGY AGENCY, Internatioanl Basic Safety Standards for Protection
Against Ionizing Radiation and for the Safety of Radiation Sources. Safety Series No.115 (1996).
— Manual of Radioisotope Production, Technical Reports Series No. 63 (1966).
— Radioisotope Production, IAEA TECDOC Series No. 125 (1970).
— Radioisotope Production and Quality Control, Technical Reports Series No. 128 (1971).
— Manual for Reactor Produced Radioisotopes, IAEA TECDOC Series No. 1340 (2003).
— Review and Assessment of Nuclear Facilities by the Regulatory Body Safety Guide, Safety Standards
Series No. GS-G-1.2 (2002).
— Safe Handling of Radionuclides, IAEA Safety Standards, Safety Series No.1 (1973).
— Production Technologies for Molybdenum-99 and Technetium-99m, IAEA-TECDOC-1065 (1999).
COMMISSION OF THE EUROPEAN COMMUNITY, Good Manufacturing Practice for Medicinal
Products in the European Community, EC (1992).
PHARMACEUTICAL INSPECTION CONVENTION, Guide to Good Manufacturing Practice for
Medicinal Products, PIC/S (2001).
PHARMACEUTICAL INSPECTION CONVENTION, Recommendations on the Inspection of Isolator
Technology, (1997).
41
PRESENTATIONS
AUTOMATION SYSTEMS FOR RADIOISOTOPE LABORATORIES
D. NOVOTNY
Hans Waelischmiller GmbH,
Dresden, Germany
Abstract
For more than 50 years the company Hans Waelischmiller GmbH (HWM) has worked in the field of
nuclear technology worldwide and designed and manufactured equipment for nuclear installations as well as
complete turnkey projects. This report deals with the activity of HWM in the field of production of radioisotopes
and radiopharmaceuticals as well as in the handling of radioactive materials in nuclear medicine departments in
hospitals.
Introduction
The company HWM is engaged in supplying complete turnkey production lines and equipment
components for radionuclides and radioactive pharmaceuticals. Some of the product lines that are
useful for radioisotope handling are given below.
ʊ Complete turnkey production lines for different radionuclides, diverse radiopharmaceuticals,
especially for
131
I labeled products as well as for the
99
Mo-
99m
Tc sterile generators
ʊ Complete turnkey PET-centers including radiopharmaceutical laboratories for preparation and
quality control, PET-camera, high-performance computer and software for data processing and
visualizing procedures as well as for other medical equipment such as radioactivity
measurement instruments and dose rate meters
ʊ Completely equipped nuclear medicine laboratories
ʊ Separate PET- cells (hermetic cells welded of stainless steel sheets and adequately shielded by
lead brick construction with liftable/lowerable shielding doors)
ʊ Air conditioning and ventilation systems with highly efficient air filters to retain any airborne
radioactive contamination (delay pipes, cooling trap or exhaust air filters).
HWM supplies hot cells for the production of radioisotopes and radiopharmaceuticals. The hot
cells are equipped with the necessary in-air and exhaust air filter systems, lead brick shielding with
steel frame including lead glass windows, locks and control panels. The hot cells are tight according to
the international standards. The construction material is stainless steel and polymeric materials. The
walls of the hot cells are smooth, impervious and unbroken. The corners are round. The hot cells
constructed for the production of radiopharmaceuticals meet the requirements for a negative pressure
isolator and GMP.
Various manipulators are used for the handling of the radioactive material inside the cell. In all
cases the manipulators are installed absolutely tight. The ball tong manipulators can be used up to
shielding thickness of 100 mm to 150 mm lead. Different master slave manipulators are available for
the handling of the radioactive material inside the cell. The master slave manipulators are used if the
shielding thickness is greater than 100 mm to 150 mm or very complicated and sensitive operations
take place inside the cell. The booting material of the manipulators is resistant to the agents used in the
cell.
HWM supplies complete in cell production equipment for various radionuclides and radioactive
pharmaceuticals as well as for complete turnkey facilities.
45

FACILITY FOR PRODUCTION OF Na
131
I -SOLUTION
The production of Na
131
I by dry distillation and the dispensing of the Na
131
I solution are carried
out in remote controlled conditions in two lead shielded hot cells. The irradiated TeO
2
-powder is
placed in a quartz furnace located in a closed furnace casing and heated up to approximately 700 °C.
The volatilized
131
I as iodine is transported in an air stream over a separator for adsorption of dust to
two so-called winding traps. The released iodine will be adsorbed in a weakly alkaline, aqueous buffer
solution with high efficiency. The process takes about 1 hour per batch. Na
131
I solution is obtained
with a very high radioactive concentration (50 to 500 GBq/ml are possible), which can be processed
for producing other radiopharmaceuticals such as capsules for diagnostic or therapeutic application
(Figs 1–2).
FIG. 1. Turnkey production facility for Na
131
I solution.
46

FIG. 2. Na
131
I production facility – in-cell equipment with furnace casing,
separator and iodine winding trap.
The hot cells are shielded with 100 mm lead and equipped with HEPA and charcoal filters for
the in-air and exhaust air. The facility has three safety barriers and safety air cycles to ensure that no
radioactive iodine can be released into the environment. The 3 barriers are as follows:
ʊ First barrier: Furnace casing, separator, iodine trap, trap pump
ʊ Second barrier: Furnace casing with air inlet and back pressure valve, process charcoal filter,
process filter pump
ʊ Third barrier: Hermetic stainless steel hot cell with lead shielding, in-air and exhaust air filters
(combination of HEPA and charcoal filter) and ventilation system of the building.
It is possible to install in the exhaust air duct an additional redundant changeable charcoal filter
system on top of the cell.
FACILITY FOR PRODUCTION OF Na
131
I CAPSULES
The production facility consists of a non-shielded glove box and a hot cell shielded with 50 mm
lead wall. Inside the glove box, the inactive upper and under parts of the gelatin capsules are put in
special pallets (5 pieces per pallet). The under part is filled with a special salt mixture. The prepared
pallets will then be transferred in the shielded production cell (Figs 3–4). Na
131
I bulk solution (in µl-
range) is transferred through the bottom port into the shielded cell. The dispensing of the necessary
Na
131
I solution into the capsule under part is carried out by a special Eppendorf-pipette moved
vertically and horizontally by linear drivers.
47

After filling the capsule under parts, the pallet with the capsule upper parts are placed over the
capsule pallet and the capsules are closed pneumatically by a semi automatic system. After closing the
capsules, the radioactivity content of each capsule is measured and the capsules are packed in vials.
FIG. 3. In-cell equipment for Na
131
I capsule production (Capsules for diagnostic and therapeutic
applications).
FIG. 4. In-cell equipment for Na
131
I capsule production
(Capsules for diagnostic and therapeutic applications).
48
FACILITY FOR PRODUCTION OF
99
Mo-
99m
Tc STERILE GENERATORS
The production facility for sterile generators consists of two shielded boxes, a suitable equipped
clean room arrangement and a packing area. The generator is a chromatographic and dry type. The
loading process with
99
Mo is the same as the elution process.
The production process is carried out in the following steps:
ʊ Preparation of all generator components;
ʊ Inactive and aseptic assembling of the generator and inactive function control;
ʊ Preparation of the
99
Mo-loading solution, dispensing of the loading portion in vials, steam
sterilization of the vials with the loading solution. This preparation of the loading solution will
be carried out in the first shielded hot cell;
ʊ Transport of the inactive and aseptic assembled generators to the loading hot cell by a transport
conveyor, which meets the pharmaceutical requirements.
Loading the generators with vials, which contain the loading solution and saline solution for
washing and evacuating vials according to the elution procedure of such a generator. The loading
procedure takes place in the second shielded box which meets the requirements of a negative pressure
isolator for pharmaceutical production. This loading procedure is used for production facilities with
batch sizes up to 50 generators per week. For higher batch sizes it is recommended that the loading
process be performed by an automatic procedure including sterile filtration.
After finishing the loading process the generator is transferred by a conventional conveyor to
the packing room. The packing process is a semi - automatic process to avoid high radiation exposure
to the staff and heavy manual works.
The advantages of this technology are the following:
ʊ Aseptic assembling of the generator is carried out inactively with an inactive quality control
ʊ Handling of the radioactivity is as simple as possible and can be carried out according to GMP-
regulation
ʊ Handling of the generator in the hospital or medical practice is simple and reliable.
HWM supplies the diverse components for the PET-technology and complete turnkey projects
in this field. The supplied components for the PET-technology meet the requirements of GMP. HWM
supplies PET-cells in big and small design with sufficient lead shielding and lead glass windows. The
cells are equipped with or without manipulators. The incorporation of the necessary clean room class
into the cells is possible. The cells can be supplied with rear or/and front walls. For special purposes
the supply of so-called mini cells is offered. The mini cells are shielded but not equipped with
manipulators. The mini cells are especially used in the PET-technology for the operation of PET-
modules and for the dispensing of bulk solutions in patient portions in vials or syringes. HWM equips
laboratories with the necessary clean room technology, ventilation systems, media supply, laboratory
furniture, devices for quality control and storage of radiopharmaceuticals. In connection with other
partners, HWM managed the installation of complete turnkey projects in the field of PET-technology.
HWM also manufactures diverse shielding components such as:
ʊ Laboratory table with table shielding and activity meter
ʊ Laboratory furniture
ʊ Lead shielded storage safes, refrigerators
ʊ Compatible shielding and moveable shielding
ʊ Tools for remote handling
ʊ Distance tools.
49
ADVANCES IN RADIOISOTOPE HANDLING FACILITIES AND
AUTOMATION OF RADIOISOTOPE PRODUCTION
L. BARANYAI
Institute of Isotopes Co. Ltd,
Budapest, Hungary
Abstract
Founded in 1959, the Institute of Isotopes of the Hungarian Academy of Sciences began to produce
radioactive isotopes in 1964. Since then, it has become a major Hungarian centre of research, development and
production relating to the application of radioisotopes. Since 1993 a part of the former Institute has been
operating as the Institute of Isotopes Co., Ltd. The main advances in radioisotope handling facilities and
automation of radioisotope production are presented here.
INTRODUCTION
The Institute of Isotopes is the major radioisotope producer of radiochemicals and
radioipharmaceuticals in Hungary. It is divided to four business units:
ʊ Radiopharmaceutical Business Branch
ʊ Immunoassay Business Branch
ʊ Molecular Biology Business Branch
ʊ Radiation Technology Business Branch.
Beside the professional branches, the Sales and Purchase Branch distributes the Company’s
products and services in the domestic and export markets.
PRODUCTION OF OPEN RADIOISOTOPES
Neutron irradiation based radioisotopes (reactor isotopes) are produced in the form of
radiochemicals (
131
I,
125
I,
32
P,
35
S,
51
Cr) and radioactive pharmaceuticals (
131
I,
153
Sm,
186
Re,
90
Y)
depending on the processing conditions and product characteristics. Fission produced
99
Mo is imported
in bulk form and is processed to Tc-generator.
Charged particle irradiation based radioisotopes (cyclotron isotopes) are purchased in bulk form
for processing to radioactive pharmaceuticals (
201
Tl,
67
Ga).
Main technological processes range from common chemical treatments to radiochemical
separation techniques (e.g. ion-exchange, liquid chromatography, distillation, extraction etc.). These
operations are supplemented by adjustment of the radioactive concentration with controlled dilution
followed by dispensing the stock solution.
Quality control methods used for checking open radioisotopes include measurement of:
radioactivity with ion-chamber based dose calibrators; radionuclidic purity by gamma ray
spectroscopy, and radiochemical purity by TLC, HPLC, electrophoresis as well as control of the
microbiological purity by growth on culture medium and LAL test.
The Company also produces labelled compounds (m
131
IBG;
32
P and
35
S-nucleotides; RIA kits
using
125
I;
14
C and
3
H-labelled organic compounds as well as undertaking custom synthesis.
51
TECHNETIUM GENERATOR PRODUCTION LINE
The ‘workhorse’ of nuclear medicine, the
99
Mo-
99m
Tc elution generator based on carrier-free
fission molybdenum is produced weekly under the licence of Amersham-Sorin in the form of contract
manufacture. The generator provides high radioactive concentration of pertechnetate solution (over 5
GBq/ml with 8 days of pre-calibration) when eluted from a ‘dry’ type alumina column. Fission
99
Mo is
imported in bulk form. Processing steps are chemical pre-treatment (oxidation) of the bulk,
preparation of stock solution by dilution, adjustment and checking of the radioactive concentration,
dispensing portions to alumina columns, testing elution on each generator and performing quality
control of the required parameters.
Bulk processing and stock solution preparation is carried out in hot cells included in class 10000
(C category) clean room environment (as the eluted
99m
TcO
4
-
solution passes through a built-in
bacteria filter, it is considered a ‘terminally sterilized product’). The generator assembling is
performed in a local class 100 (A category) clean room environment because injection needles are
unprotected here.
Peristaltic pumps are used to transfer solutions within and between the hot cells and for
dispensing portions of the stock solution using a balance for precise mass measurement. Loading of
99
Mo in to the column is done by sucking portions of the stock solution using a vacuum system. The
column and tubes are connected by nozzles.
A rolling conveyor system transfers the filled generators to the QC lab for test elutions and
further on to the package area and expedition.
CYCLOTRON AND FDG MODULE
A GE PetTrace type cyclotron and an automatic FDG panel is under installation partly with
IAEA support. The cyclotron is applicable for proton energy of 18 MeV with a maximum beam
current of 75 µA which is suitable for producing PET radioisotopes.
The Tracer lab type automatic FDG panel (synthesis module) that is controlled by computer is
used for
18
F-FDG synthesis. The actual steps of synthesis are displayed on a monitor.
18
F-FDG synthesis steps are as follows:
18
F separation from the irradiated target (
18
O enriched
water), introduction of the
18
F into organic phase through phase transfer catalyst, fluorination of the
FDG precursor (mannose-triflate) by nucleophilic substitution, hydrolysis of the protecting groups by
acidic or alkaline treatment, separation of the
18
F-FDG from the reaction mixture by a series of
cartridges. A dispenser operating under aseptic conditions supplements the FDG production system.
PRODUCTION OF SEALED SOURCES
Sealed sources are produced for industrial (
192
Ir for radiography,
60
Co for gamma irradiators)
and medical (
192
Ir for brachytherapy,
60
Co for teletherapy) applications.
192
Ir pellets with diameter of 2
mm and thickness of 0.3 mm are assembled with the required activity and welded into capsules for
industrial sources. They have specific activity about 12 TBq/g.
192
Ir sources for brachytherapy are
produced with pellet diameter of 0.5 mm and thickness of 0.035 mm and specific activity of 20 TBq/g.
60
Co sealed sources are produced with a specific activity of 0.7-0.8 TBq/g.
Contract irradiation of Ir pellets is carried out in high flux reactors (2x10
15
n.cm-
2
.sec
-1
) abroad
while long-term Co irradiations are executed in special irradiation channels of the national nuclear
power plant.
52
192
Ir sealed sources are produced in the hot cell line for sealed sources of the Company.
60
Co
sealed sources are produced in the hot cells of the gamma irradiation facility.
Electric arc welding machine (welding current 14-20 A, welding speed 4-6 mm/s, rotation speed
of 20 RPM, electrode distance 0.75 mm) is used for sealing industrial sources and a laser welding
machine (Plazmafix 50E model, welding current 1.5 A, plasma gas flow 0.3 l/min, protecting gas flow
8 l/min, welding time 0.8 s) is used for sealing brachytherapy sources. An automatic tool is used for
clamping and rotating the source under welding. Activity measurement is carried out in an ionisation
chamber. Leakage tests are performed after sealing in ultrasonic bath using nitric acid.
RADIOISOTOPE HANDLING FACILITY
The radioisotope production building licensed to nuclear ‘A’ level allowing processing of high-
levels of radioisotopes is located nearby the nuclear research reactor that have power of 20 MW and
neutron flux of 1x10
14
n.cm
-2
.s
-1
. Irradiated targets from the research reactor are transported to the
radioisotope production building in big lead containers by truck. The truck can enter into the ground-
level of the production building from where the containers are lifted up to the production level by
means of cranes.
Cranes with the container run through the receipt area and stop above the first hot cell of the
line. The container is placed into the first (called opening cell) where the target is taken out by means
of manipulators. Then the target is placed onto the trolley for transporting to the processing hot cell.
The production building includes five hot cell lines (consisting altogether of 34 hot cells) for
processing irradiated targets, producing radiochemicals, bulks for radioactive pharmaceuticals,
injectable radioactive pharmaceuticals,
99m
Tc generators and radioactive sealed sources.
Hot cells are supplied with exhaust system leading to the reactor chimney through filters. A
trolley, the position and running of which is controlled electronically, runs behind the hot cells and
carries the targets and accessories from the input cell to the individual processing cells and also the
products to the output. The governing principle is that each technology is installed in separate hot cells
in order to avoid cross-contamination. However, the hot cells are connected to each other through the
trolley tunnel so potential cross-contamination can occur through the air. Rate of the cross-
contamination is checked and evaluated by validation and is negligible.
Radiopharmaceutical and Tc generator processing hot cells are placed in clean rooms. Cold kits
are prepared in separate clean rooms supplied by air through a HVAC systems and are also exhausted
by fans forwarding the air into the reactor chimney.
Radioisotope products leave the processing hot cell lines through the outlet door located at end
of the line. Filled up vials are placed into product containers and after tinning they are transferred to
the expedition area. From this, the packed products are transferred to the radioisotope warehouse
located in the ground floor of the building and are transported to customers.
The radioisotope production area can be entered and left through a radiation monitored gate
which checks potential radioactive contamination of the operators.
HOT CELL DEVICES
Equipment and devices operating in the hot cells have been designed for manipulator operation.
Matching to the limited space within the hot cells they are compact in size. Each hot cell equipment
and device is designed locally and prepared by the Company’s workshop.
53
The personnel of the workshop carry out the maintenance of the hot cells. Several of these type
of hot cells are provided to other countries through the IAEA.
Equipment and devices designed specifically for hot cell operations are described below:
Target crushing device
It is a pneumatically operated crusher to disintegrate the irradiated target materials (e.g.
tellurium dioxide) to granules within the aluminium irradiation capsule. The capsule is placed under
the plug and then it is operated pneumatically beating the aluminium capsule mechanically.
Target cutting device
It consists of two cylinders rotated by an electric motor and a cutting disc. Aluminium capsule is
laid on the cylinders and the cutting disc is pushed to the aluminium capsule while rotating.
Ampoule capping device
A pneumatically operated crimper, the ampoule with its cap is placed on its holder by
manipulator and then the pneumatic sealing head is operated.
125
I dry distillation equipment
It consists of a compact electric oven for heating up the irradiated aluminium capsule, acid
scrubber and alkaline absorbers. The oven is temperature controlled and values are displayed.
131
I-dry distillation equipment
A compact electric oven for heating the irradiated tellurium oxide granules contained in a
silicium-carbide ceramic pot, followed by an acidic scrubber and alkaline absorbers. Temperature
controlled and supplied with a built-in activity meter.
Autoclave
A small-sized, manipulator-operated sterilisation chamber with two trays for vials, the
temperature and pressure of which can be controlled. Operating data are displayed on a screen.
Qualified for test bacteria.
Welding machine
Operable in hot cell and have welding seam control optical system for manufacturing and
quality control of sealed sources.
CLEAN ROOMS AND ISOLATORS
Natural environment is highly infected with microorganisms (its magnitude is around 10
6
cfu/surface unit). At the same time injectable pharmaceuticals must be sterile. Microorganisms
contaminating the product can be eliminated by autoclaving (product sterilisation) but metabolic
54
products of bacteria and killed bacteria (pyrogens) remain in the product and cause temperature
elevation in patients. To eliminate this, operations must be performed in spaces with low
microbiological contamination (10
-3
cfu/surface unit), called aseptic surrounding (cfu = colony
forming units).
According to the latest requirements relating to the pharmaceutical industry injectable
pharmaceuticals must be produced in aseptic space in order to protect products from bacterial and
pyrogen contamination. Clean rooms (large aseptic spaces) and isolators (local aseptic spaces)
continuously supplied with filtered air flow are used for this purpose. For radioactive products
(radioactive pharmaceuticals) prevention from radioactive contamination as well as radiation
protection aspects must also be taken into consideration. As mechanism of the biological and the
radioactive contamination is opposite to each other (biological contamination spreads from the natural
surrounding towards the product while the radioactive contamination spreads from the product
towards the surrounding) and radiation protection aspects often crosses these – harmonisation of the
three aspects and requirements is not easy.
Protection of the surrounding from radioactive contamination needs depression in the
production space while that from biological contamination needs continuous air flow around the
product. The latter is provided in the pharmaceutical industry in clean rooms where overpressure is
maintained. Combination of the two systems for radioactive products can be solved in two ways:
Either exhausted hot cells are placed into clean room (double space system) or negative pressure
isolators (single space system) are used. Negative pressure isolators are hermetically sealed aseptic
chambers that are exhausted while the inlet air is filtered. Shielded isolators provide protection against
radiation, radioisotope contamination of the surrounding and bacterial contamination of the product
within one space and are much cheaper than clean rooms.
As radioactive pharmaceuticals are generally ‘terminally sterilized products’ (they are
autoclaved), clean rooms or isolators of class 10 000 can satisfy pharmaceutical requirements. (Clean
room classes with limits of suspended particles and microorganisms in the air are found in GMP
guidelines.) However, in critical places (e.g. open injection needles at Tc-generator production) class
100 is needed.
Air circulation is provided by HVAC systems using HEPA filters for eliminating fine particles
and microorganisms from the air. Recirculation for radioactive products is not recommended. Forced
air exchange (>20 per hour) is required for the effective rinsing because in stagnating air
microorganisms would grow. Linear velocity of the introduced air is limited to the laminar range (0.3-
0.45 m/s) because turbulence would cause contamination. Temperature and air humidity are also
prescribed for clean rooms.
In addition to the clean room operation parameters many aspects relating to the design and
construction materials must be taken into consideration when designing and installing clean rooms.
As human beings also represent high risk of bacterial contamination to the product, total body
coverage (dress, mask, boot) must be worn. While operators stay in the clean room during production,
they are excluded from isolators so isolators are more beneficial from this point of view.
Surfaces of the aseptic spaces (clean rooms and isolators) must be regularly sanitized. Because
sealed spaces are difficult to clean and sanitize, application of evaporated agents (e.g. hydrogen
peroxide gas generators) have high potential in isolators and hot cells operated according to the
isolator principle. Hydrogen peroxide gas is not only very effective sanitizing agent but can penetrate
into hidden spaces not accessible for mechanical cleaning. Clean spaces must be kept under permanent
particle and microbiological control.
For smaller companies producing radioactive pharmaceuticals with low or medium capacity, the
hot cells constructed and operated according to the negative pressure isolator principle (as local clean
55
rooms) seems to be a cost saving solution. They must be totally leak-tight and supplied with air locks
for the inlet and outlet. However, their independent operation is questioned by the latest guidelines
requiring class 100 000 environment for isolators.
The Institute of Isotopes Co., Ltd. has made considerable investments to provide aseptic
environment for the preparation of radiopharmaceutical solutions (in class 10 000 clean room) with
isolator-like hot cells, Tc-generators (in class 10 000 clean room with class 100 local space) and cold
kits (in class 100 clean room).
SHARING TECHNOLOGIES TO ASEPTIC AND NON-ASEPTIC OPERATIONS
Radioisotope handling facilities consist of generally big spaces having ‘dirty’ machines (e.g.
cranes) and tons of lead containers and operating heavy hot cells serving for radiation protection. Such
systems basically differ from pharmaceutical ampoule-filling plants for which pharmaceutical
requirements have been primarily developed.
Also construction of clean rooms with continuous air flow circulation and filtering system is
very costly. Contrary to the conventional pharmaceutical industry recirculation of the filtered air is not
recommended for radioactive products, consequently much higher air flow rates need to be filtered
and conditioned increasing the operating cost. As additional drawback lead containers (‘packaging
materials’) need to be introduced into aseptic spaces increasing the risk of microbiological
contamination and the cost of protection against it.
For the above reasons small and medium sized radioisotope production companies should
carefully select critical operations which need aseptic clean spaces and plan to reduce the space of
clean rooms in order to reduce the cost of construction and operation. They should also need to
compromise several arrangements in order to harmonize nuclear (protection against radioactive
contamination and radiation of the surrounding) and pharmaceutical (protection against
microbiological contamination of the product) requirements. Also careful selection of the clean room
category (classes A, B, C, D) is important because the cost difference in differences in their
construction is not significant, but the operation and quality control costs are significantly different
e.g. according to the latest guidelines class 100 spaces need continuous particle number monitoring.
Production of radioisotopes has some further features that are not in harmony with conventional
pharmaceutical schemes. For instance the yielded activity after irradiation cannot be predicted with
high precision that would be needed for describing precise composition of the components. Also
operations such as target processing need ‘dirty’ mechanical operations and devices (e.g. capsule
crushing, cutting) as well as technological steps consisting of rough chemical and physical operations
(dry distillation of granules, dissolution of solid target materials in acids, etc.) compared to the
injection ampoule filling operation in conventional pharmaceutical industry. Due to the short half-life
of radioisotopes frequently retrospective quality control must be allowed.
Considering all these factors and constraints, the Institute of Isotopes Co. Ltd. found a
compromised solution of sharing the radiopharmaceutical production technologies to non-aseptic
target processing operations executed in traditional hot cells resulting in low volume ‘bulk’ solutions
and to aseptic bulk diluting and dispensing operations executed in clean room.
Bulk solutions are autoclaved prior to entering into the clean room. With this living
microorganisms are eliminated but pyrogen concentration can exceed the limit. However, the
subsequent dilution carried out in the aseptic clean room decreases pyrogen concentration
considerably because of the high dilution rate (>100 times) and because sterile and pyrogen-free
distilled water is used for dilution. The resultant radioactive solution is bacteria-free and its pyrogen
concentration is below the acceptance limit for injectable radioactive pharmaceuticals.
56
Based on this compromise a relatively small sized aseptic laboratory (class 10 000 clean room)
with small hot cells has been installed for dilution and dispensing operations for production of
radioactive pharmaceuticals.
GMP COMPLIANCE IN THE PRODUCTION OF RADIOACTIVE PHARMACEUTICALS
While clean rooms and isolators provide technical conditions for the aseptic production of
radioactive pharmaceuticals, regulations governing production conditions of medicinal products, i.e.
GMP, require further arrangements to be complied with for licensing such production technologies
and products.
In GMP guidelines a separate section entitled ‘Manufacture of Radioactive Pharmaceuticals’
deals with radioactive materials declaring that they are potentially hazardous, radiation protection is
needed, they have the risk of radioactive contamination, small batch sizes are generally applied, early
release is necessary due to short half-life, attention must be paid to cross-contamination and retention
of radionuclide contaminants as well as to waste disposal. At the same time injectable radioactive
pharmaceuticals must meet requirements relating to the ‘Manufacture of Sterile Medicinal Products’.
GMP guidelines relating to radioactive pharmaceuticals require self-contained facilities, lower
air pressure than surrounding to prevent radioactive contamination, product protection from
environmental contamination and air extraction without re-circulation. They emphasize importance of
process validation, equipment qualification, control and monitoring, require product release based on
written procedures and retention of reference samples.
The Institute of Isotopes Co., Ltd. devoted in the last decade considerable efforts in developing
GMP conformity and gaining production licence and registration for its radiopharmaceutical products.
Full documentation system providing traceability have been developed including site master
file, drug master files for the individual products, validation master file, specifications for materials,
manufacturing formulas and processing instructions, packaging instructions, batch processing records,
batch packaging records and several other instructions and records for such operations as receipt,
sampling, testing, release, rejection, recall, calibration, maintenance, cleaning, sanitation, treatment of
complaints, behaviour of personnel, and environmental monitoring.
Based on the technical arrangements and investments (aseptic production in clean rooms and
isolators), organisational arrangements and development of the required documentation of the
Company has reached compliance with the GMP guidelines and preconditions of licensing of the
production of injectable medicinal products. As a result of these efforts the pharmaceutical production
line and the Tc-generator production line (as well as the cold Tc-kit production laboratory) have been
accepted as complying with the GMP requirements by the national pharmaceutical authority and
production licence has been granted.
57
THE PRESENT STATUS OF HOT CELL DESIGNS AT NECSA
M. SMITH
Nuclear Energy Corporation of South Africa Ltd,
Pretoria, Transvaal, South Africa
Abstract
The radioisotope production group of the South African Nuclear Energy Corporation has manufactured a
range of radioisotopes since 1967. A 20 MW nuclear reactor is used for radioisotope production. It has modern
hot cells and an efficient waste handling facility. There are two sections dealing with radioisotope and
radiopharmaceutical production and quality control. The Radiochemical Section produces radioactive bulk
products such as
99
Mo,
32
P,
35
S and
131
I, whereas another section manufactures final products such as
131
I
diagnostic and therapeutic capsules,
192
Ir radiography sources,
137
Cs and
60
Co sealed sources,
85
Kr glass tubes for
smoke detectors. The
99
Mo-
99
Tc generators produced from
99
Mo and the complimentary kits are largely used as
radiopharmaceuticals for nuclear medicine.
INTRODUCTION
The Nuclear Energy Corporation of South Africa (NECSA) is well equipped for producing
radioisotopes. The on-site workshop, which was built to accommodate the size and requirements of the
enrichment of uranium plant, are able to produce hot cells, isotope production equipment, irradiation
rigs and transport containers. The last mentioned is helped by the plentiful supply of depleted uranium,
which is casted and machined to the required shapes.
The design of the hot cells is based on the principles of radiation protection which envisages
radiation levels below 2.5 µSvh
-1
, a free standing box within the lead-brick biological shield, the La-
Calhene DPTE transfer system for waste and sample transfers and in-cell equipment that is able to be
maintained and replaced using only the existing handling equipment. This is achieved without the
integrity of either the ventilation or shielding of the facility being compromised.
The radiation level of 2.5 µSvh
-1
or less is to be seen as the maximum level when the activity is
up against the front wall of the cell and the reading is taken on the closest point on the outside. The
effect of this decision has been that no significant exposure has ever been received at the front of the
cells. All exposures can be traced to the packing and transport of the product. The shielding is
generally in the form of 50 mm and 100 mm lead bricks, which are covered by easy washable and
replaceable plastic-coated wallpaper. A single slate of steel, which is easily removed for cell access,
constructs the roof. Although steel slabs had been used for walls, this practice has been discontinued
due to its design inflexibility and eventual resultant cost.
PRODUCTION HOT CELL FOR FISSION PRODUCED
99
Mo
The fission produced
99
Mo is the major irradiated target for producing
99m
Tc generator. In most
cases, the amount of activity in a cell is cyclic in that raw material is introduced which is then
processed and dispatched. The production of
99
Mo is an exception to this as only a small percentage of
the total activity leaves the cell after production; the remainder being waste which is stored for later
treatment. The radiation levels stay high, therefore, especially in the first cell where the initial
separation of the fission products occurs and the effects of this radiation on the manipulator booting
and lead glass window are unacceptable. To minimize the problem, the first cell has been built with a
shielded cavity under the containment box into which the waste tanks and reaction vessels have been
placed (see sketch). The cell is built with 250 mm lead brick walls but the cavity has an effective 350
mm lead to the outside and 100 mm to the box (and windows, booting) (Fig. 1).
59
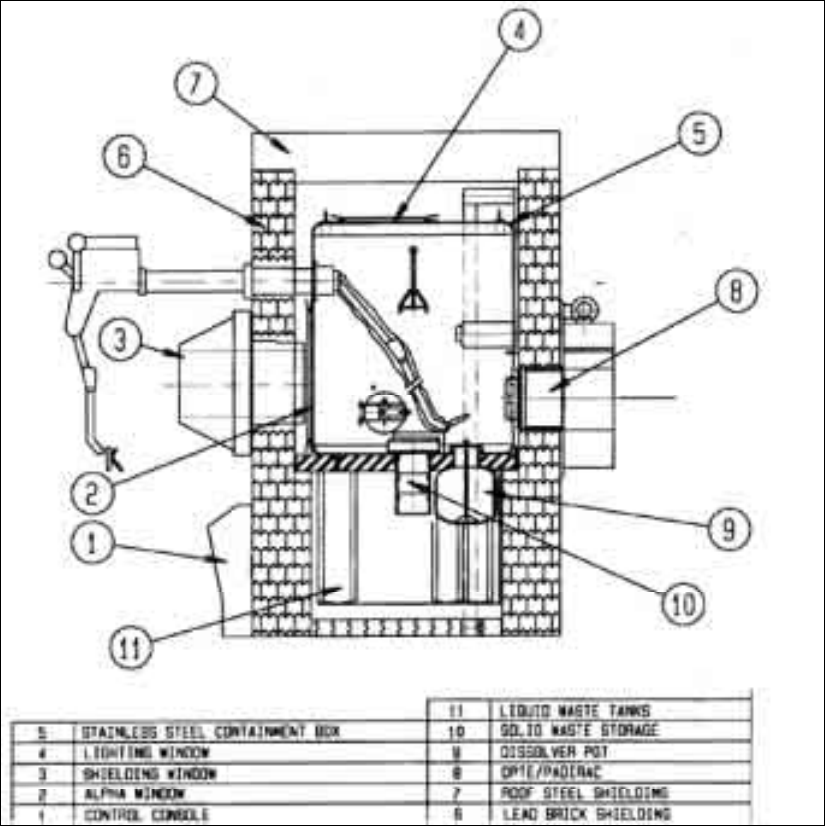
FIG. 1. Cross-section of fission produced
99
Mo production hot cell.
The freestanding boxes are constructed either with a stainless steel frame with polycarbonate
panels or with a 6 mm welded stainless steel construction and stabilized glass for viewing and lighting.
The latter is preferred for the handling of higher activities. These boxes are lowered into position
through the open roof after the walls have been built and services are introduced by access from either
side “quick disconnect” feed through.
Should a box therefore require replacement either because of maintenance reasons or to
introduce a new product facility, the radiation is reduced to an acceptable level by cleaning or decay,
the roof is removed and the box is lifted out.
The boxes are designed so that cleaning is facilitated. It has been found, however, that although
corners have been rounded and made accessible, fittings such as doors, window frames, etc, do not
receive the same attention and often form contamination traps. Improvements for this are been
continually sought.
The DPTE system from La-Calhene is extensively used in virtually all cells; in most cases the
double system is employed, which consists of concentric doors, the smaller of which is used for
sample handling and the larger for waste removal. A locally designed SARIE has been added that
allows the essentially closed DPTE system to be used to remove the product (Fig. 2).
60
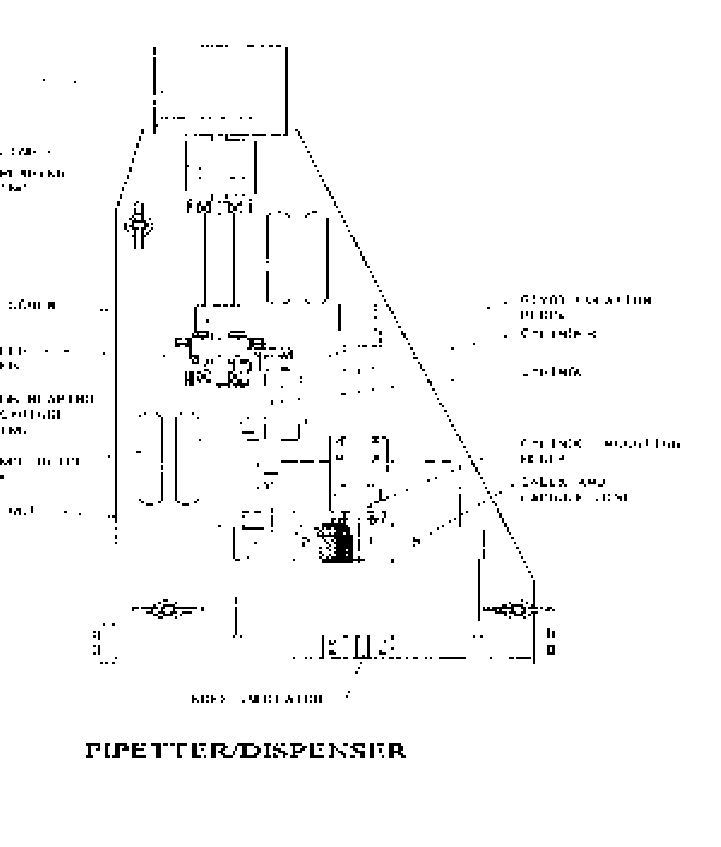
FIG. 2. Pipetter/dispenser.
The maintenance of in-cell equipment is problematic as the manipulator/tong is limited in its
mechanical capability and at times high contamination and/or radiation levels preclude access to the
box. The only safe entry is via the DPTE and a policy was adopted that all equipment be so designed
as to be removed and replaced via this system. The policy is therefore one of replacement and not of
service and repair. To this end, a storage facility was built to safely accommodate a number of the
DPTE containers, the contents of which are allowed to decay to the shape when open handling is
feasible (Fig. 3). The equipment is sent directly to the waste section should a long-lived isotope
contaminate the equipment where storage decay is impractical (Fig. 4).
61
FIG. 3. Below floor autoclave
62
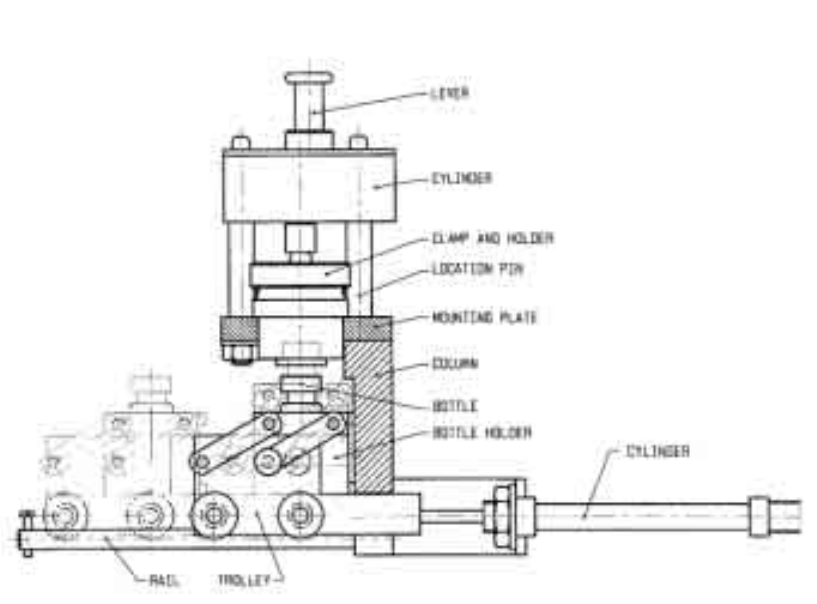
FIG. 4. Vial capper.
Although the design of hot cells has not shown much innovation over the years, it is felt that
future facilities be built around the process and not, as in the past, the process fitted into a standard
facility. In-cell equipment is being made simpler and easier to remove and replace. Further
development will also concentrate more on the requirements of pharmaceutical production.
63
LIST OF PARTICIPANTS
Baranyai, L. Institute of Isotopes Co. Ltd, Budapest, Hungary
Han, H-S. Korea Atomic Energy Research Institute,
Yusung 305-600, Taejon, Republic of Korea
Mengatti, J. Comissão Nacional de Energia Nuclear,
Instituto de Pesquisas Energeticas e Nucleares,
São Paulo, Cidade Universitaria, Brazil
Narasimhan, D.V.S. International Atomic Energy Agency
Novotny, D. Wälischmiller GmbH, Dresden, Germany
Pillai, M.R.A. International Atomic Energy Agency
Smith, M. Nuclear Energy Corporation of South Africa Ltd (NECSA),
Pretoria 0001, Transvaal, South Africa
Wagner, G. Wälischmiller GmbH, D-01328 Dresden, Germany
65
04-41451
